In this article, they wanted to see how MMP-14 was correlated with resistance to genotoxic stress and response of cancer to radio- and chemotherapy in patients. They demonstrate the elevated levels of expression of MMP-14, a matrix remodeling enzyme that degrades the extracellular environment for tissue invasion, in aggressive carcinomas compared to nonmetastatic ones, as well as how patient outcome is inversely correlated with these expression levels across all types of breast cancer.


In order to test how MMP-14 affects tumor response to genotoxic stress, they induced this type of stress in the forms of radiotherapy (Ionizing radiation) and chemotherapy (Doxorubicin, a chemotherapeutic agent) in normal and knockdown cells for this metalloprotease. They found that on KD of MMP-14 the cells are more sensitized to radiation and Dox in the metastatic cell line, as well as in the catalytically dead mutant in the non-metastatic line.

To corroborate these findings, they transplanted some cells into mice to grow tumors, keeping the same experimental conditions for Dox, and measure tumor size after two months of incubation. They found that Dox treatment in conjunction with inhibition of MMP-14 expression significantly reduces tumor growth.

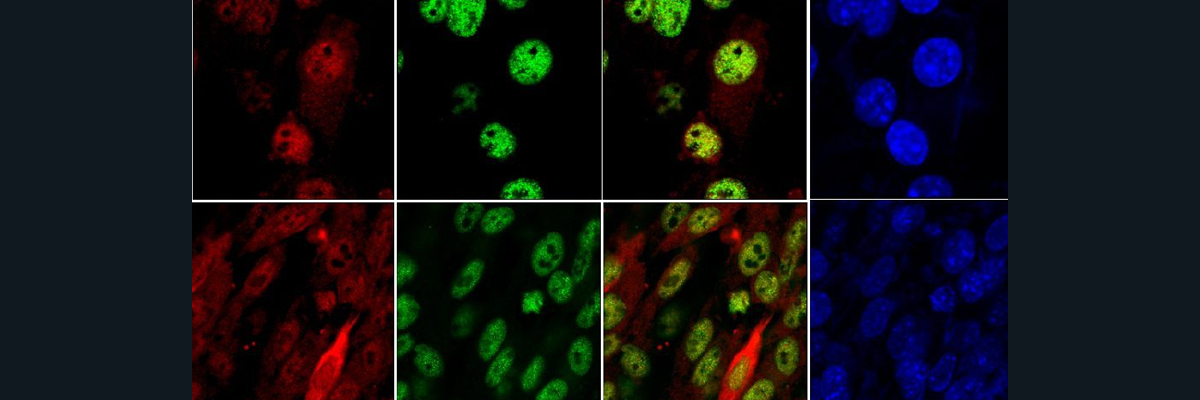
After this, they were interested in the cells’ response to DNA double strand breaks (DSBs), which are a form of major DNA damage. They quantified this damage by expression of a DNA-binding protein marker that appears when DNA needs to be repaired, and found that, after radiation, KD cells struggle more to repair their DNA damage than controls did. Additionally, they performed comet assays where they measure the tail moment of a “comet” fluorescent signal: the longer the tail, the more extensive the DNA damage. Similar to the previous experiment, they find that KD and catalytically dead mutants show an inability to cope with their DNA damage and take more time to go back to baseline levels.

They further elucidate the mechanism for this lacking DNA damage response as a defect in replication fork stability, which is essentially the machinery needed to replicate DNA and repair damage. They performed DNA fiber assays in which they stop replication by for stalling, and then allow it restart at a later time, dying those corresponding fibers as stalled (red) and restarted (green). What they found was that mutants are unable to restart replication as effectively as wild-types, showing that their incapability to repair DNA is due to replication fork instability.


The authors reason that integrin-beta receptors are correlated with ECM collagen and breast cancer, so they tested whether constitutive activation of this receptor would have an effect on DNA damage response. Not only did they find that MMP-14 silencing represses expression of integrin-beta, but more strikingly that constitutive activation of integrin-beta does in fact remove all markers for DNA damage, even on mutant cells. To test whether integrin-beta actually conferred DNA damage resistance, they performed the same experiments mentioned before with the added condition of integrin-beta constitutive activation. Survival curve in IR and Dox, tail moment, DNA fiber assay, all experiments show that integrin-beta activation confers protection from DNA damage and allow for replication fork stability and restart of DNA replication.


Citations:
Thakur, V. et al. “The membrane-tethered matrix metalloproteinase MT1-MMP triggers an outside-in DNA damage response that impacts radiotherapy responses of breast cancer”. Cancer Lett. 2019 Feb 28; 443:115-124.
https://pubmed.ncbi.nlm.nih.gov/30502358/